Dilute Solution Viscometry is one of the oldest but most informative techniques of polymer characterization. Solution viscometry reveals invaluable information on molecular density, conformation and structure (i.e. branching, chain length, chain flexibility and aggregation) of macromolecules in solution. Solution viscosity can also be used to determine the molecular weights of polymers. Intrinsic Viscosity, [η], a solution viscosity parameter, relates directly to the molecular mass of macromolecules in solution by the Mark-Houwink-Sakurada (M-H-S) equation:
[n] = KMa
Using Einstein’s viscosity equation, solution viscometry can also determine the hydrodynamic size of macromolecules in solutions:
E = E0(1+2.5V)
Intrinsic viscosity is inversely proportional to molecular density; therefore lower [η] values are indicative of higher molecular densities. For examples, two polymers, A and B, having linear and branched-chain structures, respectively, can have the same hydrodynamic size (Rh) however the molecular density will be lower for the linear polymer than the branched polymer as indicated by higher [η] for polymer A. Contrariwise, the molecular weight (M) of the branched polymer, B, will be higher than the linear polymer, A.
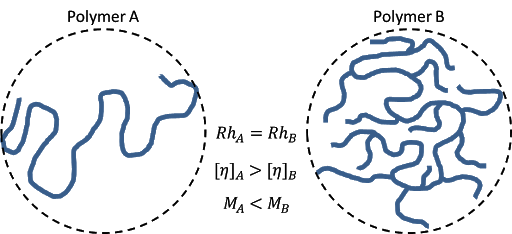
Viscosity detectors can be used online coupled with a GPC/SEC setup to determine molecular density and conformation (i.e. Hydrodynamic Volume) and therefore can be used to generate a Universal Calibration Curve. The Universal Calibration Principle is independent of the standard type and provides the true molecular weight of polymers.
Consult with our Technical Sales Specialists to discuss your application and provide you with an accurate quotation.